Laboratory of Functional Organic Compounds (N8)

Research Areas
Synthesis of organofluorine compounds
Methodology of the organic synthesis using fluorine, silicon and boron compounds
Development of C-C bond forming reactions
Main Results
Development of new nucleophilic fluoroalkylation methods
Research Group of Prof. S.L. Ioffe
Research Areas
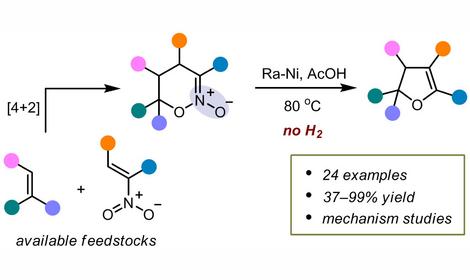
Chemistry of aliphatic nitro compounds (ANCs) and their derivatives (acyclic and cyclic nitronates), and the targeted synthesis of functional organic compounds from ANC.
Main Results
A novel strategy for the application of ANCs in the directed organic synthesis based on their silylation process has been proposed. A convenient synthesis of known highly potent Rolipram and Cilomilast analogs sourced from available precursors has been developed. The suggested synthetic strategy is considerably more efficient as compared to previous syntheses of these compounds developed by GlaxoSmithKline. The first synthesis of 1,4,6,10-tetraazaadamantanes – a novel class of heterocage compounds isomeric to Urotropin – has been accomplished.
ANC silylation is a unique process that has no close analogies in organic chemistry. It is kind of a redox process, i.e. a sequence of ANC successive transformations, in which, under the action of mediators (silylating agents), the nitro group is gradually reduced whereas the hydrocarbon skeleton is oxidized. In these transformations, protons are consecutively eliminated from α, β and γ carbon atoms of ANCs. Corresponding nitronates, N,N-bis-oxyiminium cations, nitrosoacetals, and conjugated en-oximes are intermediates in the silylation process.
The suggested approach allows one to transform a small set of available ANCs into a variety of polyfunctional derivatives using silylation as a key procedure. Many of these transformations proceed with high regio- and stereoselectivity.