Laboratory of Development and Study of Polyfunctional Catalysts (N14)

Head: Prof. Leonid M. Kustov (e-mail: lmkioc.ac.ru, tel. +7(499)137-2935)
The laboratory was established in 1954 and headed by Prof. A.M. Rubinshtein (1954-1986) and Prof. A.A. Slinkin (1986-1999).
Research Areas
Development of heterogeneous catalysts, including those supported onto novel materials (inorganic gels, metal nets, and foamed metals) and ionic liquids aimed at their use in selective oxidation of alkanes, alkenes and arenes, conversion of methane into synthesis gas, complete oxidation of trace amounts of volatile compounds in gas vents, reduction of nitrogen oxides by hydrocarbons, and removal of nitrogen oxides by NO2 adsorption.
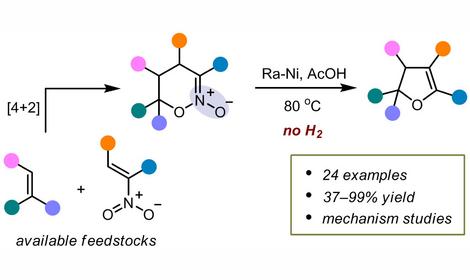
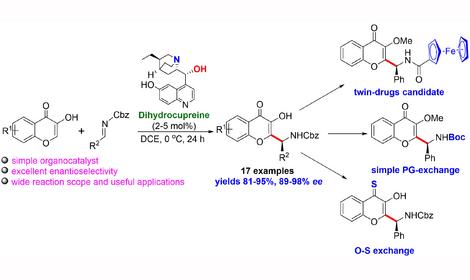
Design of heterogeneous catalysts (including sulfur-resistant ones) for hydrogenation of dienes, alkynes, N-heterocycles and aromatic compounds, ring opening in aromatic compounds, hydrodechlorination of chlorofluorohydrocarbons and CCl4, and hydrogenation of polymers; design of heterogeneous acid-base catalysts and ionic liquids for isomerization of C4-C16 alkanes, dehydroisomerization of n-butane to isobutane, nitration and alkylation of aromatic hydrocarbons and phenols, and the metathesis of cycloolefins (synthesis of a-olefins) and functionalized olefins.
Investigation of the physicochemical properties of catalysts by XPS, ESR, Fourier-IR spectroscopy of diffuse reflection, and thermosprogramed reduction and oxidation.
Studies of the catalyst activation by cold and ultrahigh frequency plasma and electron beam irradiation.
Main Results
Novel composite materials for hydrogen storage with capacity of ~8% H2 were prepared and proved to have higher efficiency and lower cost as compared to intermetallides and nanotube-based hydrogen storage systems.
It was shown that ionic liquids operating at room temperature manifested excellent performance as catalysts for oligomerization of olefins (in particular, dimerization of n-butenes, isobutylene and propylene), disproportionation and skeletal isomerization of C4-C16 paraffins, alkylation of aromatic compounds with olefins, metathesis of olefins, including functionalized ones, Gattermann-Koch carbonylation of aromatic compounds, oligomerization of benzene, biphenyl, aniline and pyrrole into oligophenylenes, oligoanilines and oligopyrroles.
Projects
Ecotoxicity of Metal and Metal Oxide Nanoparticles: Experimental Study and Modelling