Laboratory of Heterocyclic Compounds (N3)

Head: Prof. Mikhail M. Krayushkin (e-mail: mkrayioc.ac.ru, tel. +7(499)137-6939)
The laboratory had been founded in 1922 by Full Member of the Academy A.E. Chichibabin who headed it until 1931. Before merging with ZIOC, it existed as an independent Laboratory for Research and Synthesis of Vegetable and Animal Products under the Commission for the Study of Natural Productive Forces. In 1931-1945, Laboratory was headed by Professor M. M. Katznelson. Among the research fellows of Laboratory were well-known Chichibabin's associates: Full Members of the Academy I.L. Knunyants and M.I. Kabachnik, Professors N.A. Preobrazhensky and Ya.L. Goldfarb; the latter headed Laboratory in 1945-1985.
Main results (2007-2012)
Laboratory of Heterocyclic Compounds (LHC) is focused on the synthesis of photoactive compounds for optical memory. Chromone-based compounds were designed. These compounds undergo UV-induced rearrangements to form fluorescent products. The reaction is used to prepare light-sensitive media for optical data storage memory.
Methods for the synthesis of reverse photochromic compounds were developed. They are based on thermally irreversible dihetarylethenes containing thiophene and benzothiophene rings linked by cyclopentene, perfluorocyclopentene, maleimide or other heterocyclic bridges. The photochemical properties of a large series of these compounds were studied.
Dihetarylethenes with sulfur-containing functional groups, which enable their efficient binding to noble metal nanoparticles, were synthesized.
An optical device was designed based on chromones and dihetarylethenes for layer-by-layer recording, erasing, storage, and reproduction of rewritable and optical archive-type information in a multilayer (up to ten layers) recording medium.
A large series of various fulgimides, some of which exhibit fluorescence properties, were synthesized by reactions of thiophene-based fulgides with various amines and their photochromic properties were studied. Reactivity of the products and their photochromic properties were shown to be similar to those of dihetarylethenes. These compounds can be used in optical memory devices.
The Fischer reaction of thiophene derivatives has been studied systematically. The research resulted in the synthesis of previously unavailable thienopyrroles and thienopyrrolenines, which were used to design a new type of unique photochromic spiropyrans, spirooxazines, and merocyanine dyes.
Investigations of S-functionalization of organic compounds with elemental sulfur led to the enlargement of the heterocyclic synthesis scope based on simple and easily available starting compounds. Methods were elaborated for the synthesis of a wide range of monothiooxamides, oxamic acid thiohydrazides and their derivatives containing aromatic and heteroaromatic moieties, which served as a basis for the preparation of various antibacterial compounds, in particular biologically active low-toxicity substances that inhibit the type III secretion system of pathogenic bacteria.
An approach to new fused dihydropyridinone-containing heterocyclic systems was developed. It is based on three-component condensation of corresponding aminoheterocycles with aldehydes and Meldrum’s acid. Calculations with the use of the PASS program predicted that the synthesized dihydropyridines should have psychotropic, anti-neurotic, nootropic or antipsychotic activities.
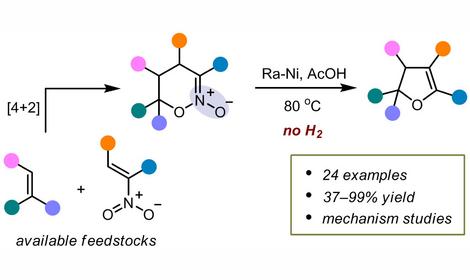
Methods for the synthesis of nitrogen-containing derivatives of aromatic heterocycles such as furazan, furoxan, 1,2,4- and 1,3,4-oxadiazoles, tetrazole, etc. under atmospheric and high pressure (10-14 kbar) were devised. The compounds were used to produce high-density polynitrogen materials. The 1,3-dipolar cycloaddition reactions were studied and original methods for the synthesis of stable aromatic nitrile oxides were developed. These methods provide the basis for a pioneering environmentally friendly technology of low-temperature vulcanization of rubber, which finds applications both in our country and abroad.
Laboratory has great competence in the synthesis of additives for tire manufacturing. A manufacturing technology was worked out for the production of the anticorching agent SANTOGARD PVI. A large series of sulfenimides was synthesized.
LHC successfully collaborates with world's largest companies such as Michelin, Dow Chemical, Intel, DuPont, Samsung, Lanxess, DuPont, Samsung, and so on.
Publications
- A. G. Lvov Switching the Mallory Reaction to Synthesis of Naphthalenes, Benzannulated Heterocycles, and Their Derivatives J. Org. Chem. 2020, 85, 8749−8759. DOI: 10.1021/acs.joc.0c00924
- V. P.Perevalov, V. S.Mityanov, B. V.Lichitsky, A. N.Komogortsev, L. G.Kuz’mina, T. Yu. Koldaeva, V. S. Miroshnikov, Anton V. Kutasevich Synthesis of highly functional imidazole derivatives via assembly of 2-unsubstituted imidazole N-oxides with CH-acids and arylglyoxals Tetrahedron 2020, 76(8), 130947. DOI: 10.1016/j.tet.2020.130947
- B. V. Lichitskii, V. G. Melekhina,A. N. Komogortsev, C. V. Milyutin, A. N. Fakhrutdinov, Y. O. Gorbunov, and M. M. Krayushkin Synthesis of substituted naphtho[1,2-b]benzofuran-7(8H)-ones via photoinduced rearrangement of 4H-chromen-4-one derivatives Org. Biomol. Chem. 2020, 18(13), 2501-2509.DOI: 10.1039/D0OB00149J
- A.Geronikaki, A.Petrou, M.G.Akrivou, I.S.Vizirianakis, F.M.Chatzopoulou, B.Lichitsky, S.Sirakanyan, M.Kostic, M.Smiljkovic, M.Soković, D.Druzhilovskiy, V.Poroikov, Antibacterial activity of griseofulvin analogues as an example of drug repurposing, Antibacterial activity of griseofulvin analogues as an example of drug repurposing, Int. J. Antimicrob. Agent, 2020, 55(3), [UNSP 105884]. DOI: 10.1016/j.ijantimicag.2020.105884
- A. V. Zakharov,A. V. Yadykov,A. G. Lvov,E. A. Mitinaand V. Z. Shirinian Photochemical rearrangement of diarylethenes: synthesis of functionalized phenanthrenes, Org. Biomol. Chem.2020, 18(16), 3098-3103. DOI: 10.1039/D0OB00296H
- A. G. Lvov, A. V. Yadykov, K. A. Lyssenko, F. W. Heinemann, V. Z. Shirinian, M. M. Khusniyarov Reversible Shifting of a Chemical Equilibrium by Light: The Case of Keto–Enol Tautomerism of a β-Ketoester, Org. Lett. 2020, 22, 2, 604–609. DOI: 10.1021/acs.orglett.9b04376
- A. G Lvov, Y. Yokoyama, V. Z Shirinian Post-Modification of the Ethene Bridge in the Rational Design of Photochromic Diarylethenes,Chem. Rec. 2020, 20(1), 51-63. DOI: 10.1002/tcr.201900015
- V. A. Migulin , A New Synthetic Pathway to Symmetric Bisubstituted Naphthoquinones, Synthesis 2020; 52(01): 60-68. DOI: 10.1055/s-0039-1690725
- A. G. Lvov, A. Bredihhin,; J. Peet, A. V. Yadykov, A. O. Dmitrienko, V. Z. Shirinian 1,2-Bis- and 1,2,3-tris(2,5-dimethylthiophen-3-yl)azulenes: Synthesis, structure and properties Dyes Pigm. 2020, 172, 107843. DOI: 10.1016/j.dyepig.2019.107843
- A. V. Yadykov,V. Z. Shirinian, Recent Advances in the Interrupted Nazarov Reaction, Adv. Synth. Catal., 2020, 362(4), 702-723.DOI: 10.1002/adsc.201901001
- A. G.Lvov, Y.Yokoyama, V. Z. Shirinian Post-modification of the ethene bridge in the rational design of photochromic diarylethenes Chem. Rec. 2020, 20(1), 51-63. DOI: 10.1002/tcr.201900015 (in collaboration with Yokohama National University, Japan).
- V.G. Melekhina, V.S. Mityanov, B.V. Lichitsky, A.N. Komogortsev, K.A. Lyssenko, M.M. Krayushkin Synthesis of Benzocarbazole Derivatives by Photocyclization Eur.J. Org. Chem., 2019, 1335-1340. DOI: 10.1002/ejoc.201801664 (in collaboration with Mendeleev University of Chemical Technology of Russia and Institute of Organoelement Compounds RAS).
- A. G. Lvov, M. Mörtel, A. V. Yadykov, F. W. Heinemann, V. Z. Shirinian, M. M. Khusniyarov Photochromic diarylethene ligands featuring 2-(imidazol-2-yl)pyridine coordination site and their iron(II) complexes Beilstein J. Org. Chem. 2019, 15, 2428–2437. DOI: 10.3762/bjoc.15.235 (in collaboration with Friedrich–Alexander University Erlangen–Nürnberg, Germany).
- A. G. Lvov, A. M. Kavun, V. V. Kachala, K. A. Lyssenko, V. Z. Shirinian, Photorearrangement of dihetarylethenes as tool for benzannulation of heterocycles Org. Biomol. Chem. 2019, 17, 4990–5000. DOI: 10.1039/c9ob00690g (in collaboration with Institute of Organoelement Compounds RAS).
- D. D. Dashitsyrenova, A. G. Lvov, L. A. Frolova, A. V. Kulikov, N. N. Dremova, V. Z. Shirinian, S. M. Aldoshin, M. M. Krayushkin, P. A. Troshin Molecular structure–electrical performance relationship for OFET-based memory elements comprising unsymmetrical photochromic diarylethenes J. Mater. Chem. C 2019, 7, 6889-6894. DOI: 10.1039/c9tc01273g (in collaboration with Institute of Problems of Chemical Physics RAS).
- A. G. Lvov, A. V. Zakharov, K. A. Lyssenko, V. V. Kachala, V. Z. Shirinian Dialkylation of ethyl 4-(het)aryl-3-oxobutanoates as a route to 5-(2-oxoethyl)cyclopentenones Synlett 2019, 30, 1321-1323. DOI: 10.1055/s-0039-1689926 (in collaboration with Institute of Organoelement Compounds RAS).
- A. V. Zakharov, A. G.Lvov, I. A. Rostovtseva, A. V. Metelitsa, A. V. Chernyshev, M. M. Krayushkin, A. V. Yadykov, V. Z. Shirinian Photocyclization of diarylethenes: the effect of imidazole on the oxidative photodegradation process Photochem. Photobiol. Sci. 2019, 18, 1101–1109. DOI: 10.1039/c8pp00507a (in collaboration with Southern Federal University).
- V. Z. Shirinian, I. A. Lonshakov, A. V. Zakharov, A. G. Lvov, M. M. Krayushkin, Practical deoxygenation of oxazole N-oxides by PCl3-collidine Synthesis 2019, 51, 414–420. DOI: 10.1055/s-0037-1610278
- V. Mityanov, V. Melekhina, A. Fakhrutdinov, V. Barachevsky, T. Valova, A. Ayt, I. Martynov, K. Lyssenko, M. Krayushkin Dihetarylethene photocyclization as a synthetic route to fluorescent compounds. J. Photochem. Photobiol. A: Chemistry 2019, 369, 34-43. DOI: 10.1016/j.jphotochem.2018.10.017 (in collaboration with Mendeleev University of Chemical Technology of Russia and Photochemistry Center RAS).
- A. G. Lvov, M. M. Khusniyarov, V. Z. Shirinian Azole-based diarylethenes as the next step towards advanced photochromic materials J. Photochem. Photobiol. C: Photochem. Rev., 2018, 36, 1-23. DOI: 10.1016/j.jphotochemrev.2018.04.002 (in collaboration with Friedrich–Alexander University Erlangen–Nürnberg, Germany).
- S. Jana, A. Chakraborty, V. Z. Shirinian, A. Hajra Synthesis of Benzo[4,5]imidazo[2,1‐b]thiazole by Copper(II)‐Catalyzed Thioamination of Nitroalkene with 1H‐Benzo[d]imidazole‐2‐thiol Adv. Synth. Catal. 2018, 360, 2402-2408. DOI: 10.1002/adsc.201800393 (in collaboration with Visva-Bharati Central University, Santiniketan, India).
- S. Jana, S. Samanta, A. K. Bagdi, V. Z. Shirinian, A. Hajra Metal-free C–H arylation of imidazoheterocycles with aryl hydrazines RSC Adv., 2018, 8, 12360-123367. DOI: 10.1039/c8ra01474d (in collaboration with Visva-Bharati Central University, Santiniketan, India).
- А. G. Lvov, A. M. Alexeeva, E. A. Lvova, M. M. Krayushkin, V. Z. Shirinian Spectral properties and structure of unsymmetrical diarylethenes based on thiazole ring with hydrogen at the reactive carbon Spectr. Acta Part A: Mol. Biomol. Spectrosc., 2018, 203, 348-356. DOI: 10.1016/j.saa.2018.05.097.
- Chapter in the book: V. Barachevsky, M. Krayushkin and V. Kiyko Light-Sensitive Organic Recording Media for Three-Dimensional Optical Memory, (Chapter 9) in Photon-Working Switches, 2017, 181-207. Ed. Springer Japan KK, Y. Yokoyama and K. Nakatani (eds.). DOI 10.1007/978-4-431-56544-4_9
- V. Z. Shirinian, , A. M. Kavun, A. G. Lvov, I. V. Zavarzin, M. M. Krayushkin Practical Efficient Synthesis of Polyaryl(hetaryl)substituted Cyclohexenones and Salicylates Synthesis 2017, 49, 1255. DOI: 10.1055/s-0036-1588908
- A. V. Zakharov, E. B. Gaeva, A. G. Lvov, A. V. Metelitsa, V. Z. Shirinian, Photochemical Rearrangement of Diarylethenes: Reaction Efficiency and Substituent Effects J. Org. Chem. 2017, 82, 8651−8661. DOI: 10.1021/acs.joc.7b01587 (in collaboration with Institute of Physical and Organic Chemistry of Southern Federal University).
- A. G. Lvov, N. Milevsky, A. M. Yanina, V. V. Kachala, V. Z. Shirinian Aerobic Dimerization of Ethyl 4‑Thienyl-3-ketobutanoate toward a Modifiable Photochromic Diarylethene Precursor Org. Lett. 2017, 19, 4395−4398. DOI: 10.1021/acs.orglett.7b02143 (in collaboration with Mendeleev University of Chemical Technology of Russia).
- V. A. Migulin, A. G. Lvov, M. M. Krayushkin, Photoisomerization of cyclopentene-based b-(2-furanyl)- and b-(2-thienyl)enones, Tetrahedron 2017, 73, 4439-4449. DOI: 10.1016/j.tet.2017.06.005
- A. G. Lvov, V. Z. Shirinian Photoinduced Rearrangements of Diarylethenes Chem. Heterocycl. Compd. 2016, 52, 658-665. DOI: 10.1007/s10593-016-1946-z
- M.L. Keshtov, S.A. Kuklin, N. Radychev, A.Y. Nikolaev, I.E. Ostapov, M M Krayushkin, I. O. Konstantinoy, E.N. Koukaras, A. Sharma, G.D. Sharma New low bandgap near-IR conjugated D-A copolymers for BHJ polymer solar cell applications. PhysChemChemPhys 2016, 18, 8389-8400. DOI: 10.1039/C5CP07705B (in collaboration with Institute of Organoelement Compounds RAS).
- K. Ramkumar, S. Samanta, A. Kyani, S. Yang, S. Tamura, E. Ziemke, J. A. Stuckey, S. Li, K. Chinnaswamy, H. Otake, B. Debnath, V. Yarovenko, J. S. Sebolt-Leopold, M. Ljungman, N. Neamati Mechanistic evaluation and transcriptional signature of a glutathione S-transferase omega 1 inhibitor Nat. Commun. 2016, 7, 13084. DOI: 10.1038/ncomms13084 (in collaboration with University of Michigan, USA).
- V. Z. Shirinian, D. V. Lonshakov, A. G. Lvov, A. M. Kavun, A. V. Yadykov, M. M. Krayushkin Photo- and PH-switchable fluorescent diarylethenes based on 2,3-diarylcyclopent-2-en-1-ones with dialkylamino groups Dyes Pigm. 2016, 124, 258-267. DOI: 10.1016/j.dyepig.2015.09.027 (in collaboration with Mendeleev University of Chemical Technology of Russia).
- L. A. Frolova, A. A. Rezvanova, V. Z. Shirinian, A. G. Lvov, A. V. Kulikov, M. M. Krayushkin, P. A. Troshin OFET-based memory devices operating via optically and electrically modulated charge separation between the semiconductor and 1,2-bis(hetaryl)ethene dielectric layers Adv. Electr. Mat. 2016, 2, 1500219. DOI: 10.1002/aelm.201500219(in collaboration with Institute of Problems of Chemical Physics of RAS).
- V. Z. Shirinian, A. G. Lvov, E. Yu. Bulich, A. V. Zakharov, M. M. Krayushkin Novel photochromic diarylethenes bearing an imidazole moiety Tetrahedron Lett. 2015, 56, 5477–5481. DOI: 10.1016/j.tetlet.2015.08.028 (in collaboration with Mendeleev University of Chemical Technology of Russia).
- A. G. Lvov, V. Z. Shirinian, A. V. Zakharov, M. M. Krayushkin, V. V Kachala, I. V. Zavarzin A general photo-induced sequential electrocyclization / [1,9]-sigmatropic rearrangement / ring-opening reaction of diarylethenes J. Org. Chem. 2015, 80, 11491−11500. DOI: 10.1021/acs.joc.5b02237
- K.S. Levchenko, V.A. Barachevski, O.I. Kobeleva, O.V. Venidiktova, T.M. Valova, A.M. Bogacheva, K.A. Chudov, E.P. Grebennikov, P.S. Shmelin, N.O. Poroshin, G.E. Adamov, V.N. Yarovenko, M.M. Krayushkin. Synthesis of new fluorescent 1-(thien-2-yl)-9H-thieno[3,4-b]-chroman-9-ones and their fluorescent photomodulation by photochromic dihetarylethenes Tetrahedron Letters, 2015, 56, 1085–1088. DOI: 10.1016/j.tetlet.2015.01.101 (in collaboration with Photochemistry Center RAS and Central Scientific Research Technological Institute “Technomash”).
- V. А. Мigulin, М. М. Кrayushkin, V. A. Barachevsky, O. I. Kobeleva, V. V. Novikov, K. A. Lyssenko Synthesis and Studies of Symmetric Dibenzothienylcyclopentenes Tetrahedron 2015, 71, 584-598. DOI: 10.1016/j.tet.2014.12.036 (in collaboration with Photochemistry Center RAS).